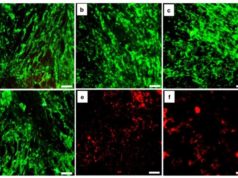
PolarityTE, a company specialised in regenerative tissue products and biomaterials, has announced findings from an open-label, single-arm pilot study, which examined the impact of SkinTE in closing venous stasis leg ulcers (VLUs) following the failure of conventional treatments.
The clinical outcomes were reported in a poster presentation, entitled “Pilot Study Assessing Novel Autologous Homologous Skin Construct Treatment of Venous Stasis Leg Ulcers”, at the Symposium on Advanced Wound Care Fall meeting (SAWC Fall 2019; 12-14 October, Las Vegas, USA).
“The completion of this pilot study is a pivotal step in further establishing SkinTE as a treatment for VLUs,” said study first author David Armstrong (Keck School of Medicine, University of Southern California, Los Angeles, USA). He added: “As a clinician and researcher, study findings provide valuable learnings into real-world outcomes and offer the opportunity to broaden treatment options for healthcare providers and patients. We look forward to further data that build upon these initial results.”
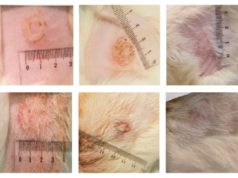
The pilot study included 10 patients with VLUs that remained open after at least one month of conventional treatments. The patients were treated with SkinTE, an autologous, homologous human cellular and tissue-based product designed to regenerate full-thickness, functional skin for the repair, reconstruction, and replacement of a patient’s own skin.
The study found an 80 percent closure rate of the VLUs within 12 weeks after treatment with SkinTE. In addition, all of the patients’ wounds demonstrated graft take and initial signs of closure, including granulation and progressive epithelialisation, shortly after a single treatment with SkinTE.
One VLU, which had previously been deemed closed, had reopened prior to the 2-week durability visit as a result of external factors unrelated to the SkinTE procedure. Furthermore, another VLU did not close within 12 weeks, which was the largest in the study (12.2cm2) and had remained open for several months prior to treatment with SkinTE after previously failed treatment with a split-thickness skin graft; this VLU closed within 13.5 weeks post application of SkinTE.
“We are pleased with the results of our VLU pilot study and today’s announcement further signifies the strides we are making to build a comprehensive clinical body of evidence to validate the utility of SkinTE,” said Nikolai Sopko, chief scientific officer of PolarityTE.
“As we look ahead, we remain committed to investing in clinical initiatives that meet the needs of patients, healthcare providers, and payors. Importantly, the goal of our VLU pilot study was to gain critical insights to design our multi-centre, randomised controlled trial, which is currently enrolling patients and will rigorously evaluate the ability of SkinTE to treat VLUs in a larger subset of patients.”