Advanced fluorescence imaging has the potential to decrease bioburden and significantly improve the healing rates of chronic wounds, according to the results of a case series investigation conducted in Cleveland, USA. As well as targeting debridement to promote wound healing, fluorescence imaging was also found to be capable of providing positive revenues in an outpatient wound care setting.
Featured in the prize-winning oral abstract session at the Symposium of Advanced Wound Care Fall meeting (SAWC Fall 2019; 12-14 September, Las Vegas, USA), presenter Windy Cole (Cleveland, USA) emphasised that an optimally prepared wound bed which promotes healing must be regularly debrided to remove devitalised tissues, while a reduction of the bacterial load is also necessary. Despite the elimination of debris and slough at the base of the wound, Cole questioned “are we really getting rid of bioburden and affecting the bacterial contamination of the wound?”.
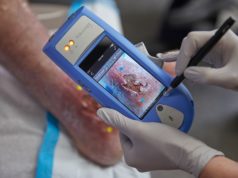
The emergence of a point-of-care fluorescence imaging system that can help clinicians with bacterial-targeted debridement and bioburden management, in the treatment of chronic wounds, is considered to be an innovation with high potential, prompting an observational case series which examined the possible benefits of using such a device.
According to Cole, 11 chronic wounds in the lower extremities were imaged weekly, as part of the investigation, with the device, highlighting both bacterial fluorescence and wound area over 12 weeks (or until either healing or lost follow-up). Once images positive for bacterial fluorescence developed, they were used to guide debridement to those specific regions.
Following this stage, data from each assessment were categorised into two groups – either a fluorescence-positive assessment or fluorescence-negative assessment – while the percentage of weekly change in slopes of wound area were also calculated from linear regressions for each category.
Ultimately, six out of the 11 wounds healed during the 12-week case series. Cole outlined that at week one, the average area of the wounds was 8.1cm², with 10 of the wounds exhibiting red fluorescence to indicate the presence of bacteria. Moreover, this bacterial fluorescence continued for 3.7 weeks on average, while no wounds begun to heal during the period in which red fluorescence remained present.
With the support of information provided by fluorescence images, it was then possible to better debride the affected area to an appropriate level, as Cole asserted when presenting the case series results. It was also revealed that in 9% of cases additional debridement resulted in a high billing code. Overall, the average weekly change in wound area increased (6.5 ± 10.8) during periods in which bacterial fluorescence was present, compared to a significant decrease (027.7 ± 10.1, p=0.048) after the targeted debridement and further treatment had successfully eliminated the bacteria.
Results of the study, as Cole confirmed, suggest that the aid of fluorescence information improved efforts to reduce the bacterial load and helped to foster a better environment for the wound to heal. Cole concluded that the findings of this case series advocate the incorporation of fluorescence guidance into standard of care as a way of boosting wound healing trajectories, improving patient outcomes and guiding clinicians to more appropriate treatments. Additionally, the increased revenues this technology can offer to outpatient wound centres also represents a significant benefit.
Cole concluded: “I feel that the incorporation of fluorescence guidance into standard of care could help to increase wound healing trajectories, improve outcomes and lead to more appropriate wound care therapy, because it can tell us whether the dressings we are applying to our patients are actually working. There are a lot of dressings and wound ointments that tell us they will reduce biofilms and bioburden, but are the really doing that? We cannot see this with our naked eye, but we can with the fluorescence imaging camera.”